Expressing Concentration of Solutions
Monitoring Water Pollution: The Role of ppm in Ensuring Safe Drinking Water
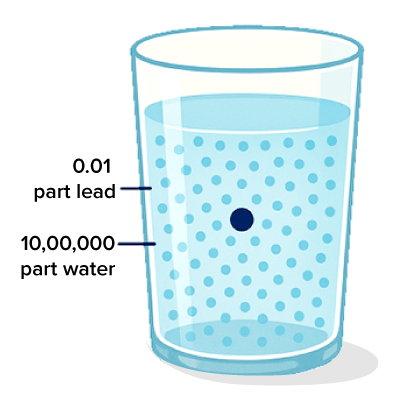
Scientists and regulatory bodies often measure the level of pollutants such as heavy metals (like lead or mercury) or harmful chemicals (like nitrates or pesticides) in water bodies using the unit parts per million (ppm). For example, the safe limit for lead in drinking water, as recommended by the Bureau of Indian Standards (BIS) standards, is 0.01 ppm. This means that in one million parts of water, only 0.01 parts of lead should be present. If the concentration exceeds this limit, the water is considered unsafe for human consumption. Using ppm allows detection and regulation of even minute quantities of harmful substances, making it a vital unit for ensuring environmental and public health safety.
Expressing Concentration of Solutions
A solution is a homogeneous mixture of two or more substances. Generally, the substance present in a smaller proportion is called the solute, and the substance present in a larger proportion is called the solvent. Solvent determines the physical state in which the solution exists. Understanding the amount of solute dissolved in a known amount of solvent or solution is crucial in various chemical processes and calculations. This amount is expressed as the concentration of the solution.
There are several methods to express the concentration of a solution including percentage concentration, strength of the solution, molarity, molality, normality, mole fraction, mass fraction, parts per million, and formality, each with its own advantages and applications.
Percentage Concentration
Percentage concentration expresses the amount of solute present in 100 parts of the solution. It can be expressed in three methods:
Percentage by Mass (w/w)
This represents the mass of the solute in grams present in 100 grams of the solution. It is calculated using the formula:
@$\begin{align*}\text{Mass % of a component (w/w)} = \frac{\text{Mass of the component in the solution}}{\text{Total mass of the solution}} \times 100\end{align*}@$
For example, a 5% solution of glucose by mass means that 5 grams of glucose are dissolved in 95 grams of water, making a total of 100 grams of the solution.
Percentage by Volume (v/v)
This represents the volume of the liquid solute in millilitres (or cubic centimetres) present in 100 millilitres (or cubic centimetres) of the solution. It is primarily used when both the solute and the solvent are liquids. The formula is:
@$\begin{align*}\text{Volume % of a component (v/v)} = \frac{\text{Volume of the component}}{\text{Total volume of the solution}} \times 100\end{align*}@$
For example, a 20% solution of ethanol in water by volume means that 20 ml of ethanol is dissolved in water to make a total volume of 100 ml of the solution.
Mass by Volume Percentage (w/v)
This represents the mass of the solute in grams dissolved in 100 millilitres (or cubic centimetres) of the solution. It is commonly used in pharmacy and medicine. The formula is:
@$\begin{align*}\text{Mass by volume % of a component (w/v)} = \frac{\text{Mass of the component in the solution (in grams)}}{\text{Total volume of the solution (in ml)}} \times 100\end{align*}@$
For example, a 0.9% saline solution (w/v), commonly used as an intravenous fluid, contains 0.9 grams of sodium chloride (NaCl) dissolved in 100 ml of the solution.
When the type of percentage is not specified, it is usually assumed to be percentage by mass (w/w).
PLIX: Percent Solutions
Strength of Solution
The strength of a solution is defined as the amount of solute in grams present in one litre (or one cubic decimeter, dm3) of the solution. It is expressed in grams per litre (g/L) or grams per cubic decimeter (g/dm3).
@$\begin{align*}\text{Strength of a solution} = \frac{\text{Mass of the solute in grams}}{\text{Volume of the solution in litres}}\end{align*}@$
For example, if 10 grams of potassium hydroxide (KOH) are dissolved in 500 ml of solution, the strength of the solution is:
@$\begin{align*}\text{Strength} = \frac{10 \text{ g}}{0.5 \text{ L}} = 20 \text{ g/L}\end{align*}@$
Molarity (M)
Molarity is one of the most commonly used units for expressing concentration in chemistry. It is defined as the number of moles of the solute dissolved per litre (or cubic decimeter) of the solution. It is denoted by the symbol 'M' and its unit is mol/L.
@$\begin{align*}\text{Molarity (M)} = \frac{\text{Number of moles of solute}}{\text{Volume of the solution in litres}}\end{align*}@$
The number of moles of a solute can be calculated by dividing the mass of the solute by its molar mass:
@$\begin{align*}\text{Number of moles of solute} = \frac{\text{Mass of solute (in grams)}}{\text{Molar mass of solute (in g/mol)}}\end{align*}@$
Combining these two equations, if 'w' grams of a solute with a molar mass 'Molar mass' are dissolved in 'V' ml of solution, the molarity can be calculated as:
@$\begin{align*}\text{M} = \frac{w}{\text{Molar mass}} \times \frac{1000}{V}\end{align*}@$
Molarity is also related to the density of the solution. Suppose w g of solute is dissolved in 100 ml of solution. If d is the density of solution in g/ml then:
@$\begin{align*}\text{Volume of solution} = \frac{\text{Mass of solution}}{\text{Density of solution}} = \frac{100}{d}\text{ml}\end{align*}@$
@$\begin{align*}\text{M} = \frac{w}{\text{Molar mass of the solute}} \times \frac{1000 \times d}{100} =\frac{w\times 10 \times d}{\text{Molar mass of the solute}} \end{align*}@$
Examples of Molarity
Example 1:
Calculate the molarity of a solution containing 4.9 grams of sulfuric acid (H2SO4) dissolved in 500 cm3 of solution.
@$\begin{align*}\text{The molar mass of} \ \ \text{H}_2\text{S}\text{O}_4 = (2 \times 1) + 32 + (4 \times 16) = 98 \ \ \text{g/mol}\end{align*}@$
@$\begin{align*}\text{M} = \frac{4.9 \text{ g}}{98 \text{ g/mol}} \times \frac{1000 \, mL/L}{500 \text{ mL}} = 0.05 \text{ mol} \times 2 \text{ L}^{-1} = 0.1 \text{ mol L}^{-1} = 0.1 \text{ M}\end{align*}@$
Example 2:
A solution contains 20% by mass of sodium hydroxide (NaOH). If the density of the solution is 1.08 g/cm³ and the molar mass of NaOH is 40 g/mol, calculate the molarity of the solution. Assume 100 g of the solution for the easy calculation as concentration is independent of the amount of the solution.
Given a 20% by mass solution of sodium hydroxide (NaOH), which means 20 g of NaOH is present in every 100 g of the solution.
To calculate molarity, we need the volume of the solution. The density of the solution is given as 1.08 g/cm³, so:
@$\begin{align*}\text{Volume of solution} = \frac{\text{Mass of solution}}{\text{Density}} = \frac{100\,\text{g}}{1.08\,\text{g/cm}^3} = \frac{100}{1.08}\text{cm}^3 = \frac{100}{1.08}\text{mL} = \frac{0.1}{1.08}\text{L}\end{align*}@$
Next, calculate the number of moles of NaOH:
@$\begin{align*}\text{Moles of NaOH} = \frac{20\,\text{g}}{40\,\text{g/mol}} = 0.5\,\text{mol}\end{align*}@$
Now, the molarity (M) is given by:
@$\begin{align*}\text{M} = \frac{\text{Moles of solute}}{\text{Volume of solution in litres}} = \frac{0.5}{\frac{0.1}{1.08}} = 5.40\,\text{mol/L}\end{align*}@$
Therefore, the molarity of the solution is 5.40 mol/L.
Alternative Method:
@$\begin{align*}\text{M} = \frac{w\times 10 \times d}{\text{Molar mass of the solute}} \end{align*}@$
@$\begin{align*}\text{M} = \frac{20\times 10 \times 1.08}{\text{40}} = 5.40 \text{mol/L}\end{align*}@$
Formality (F)
For ionic compounds that do not exist as discrete molecules in solution (e.g., NaCl, Na2CO3), the term formality (F) is used instead of molarity. Formality is defined as the number of gram formula mass (or formula units) of the solute dissolved per litre of the solution. The formula mass is the sum of the atomic masses of the atoms in the formula of the compound. For most practical purposes in dilute solutions, formality and molarity have the same numerical value.
For example, a 0.2 F aqueous solution of sodium carbonate (Na2CO3) contains 0.2 gram formula masses of Na2CO3 per litre of solution. In this solution, one mole of Na2CO3 dissociates to give two moles of sodium ions (Na+) and one mole of carbonate ions (CO32-). Therefore, a 0.2 F Na2CO3 solution is also 0.4 M in Na+ ions and 0.2 M in CO32- ions. So, while the formality of the Na2CO3 solution is 0.2 F (referring to the concentration of the undissociated formula units), the molarities of the individual ions in the solution are different due to the stoichiometry of the dissociation.
Normality (N)
Normality is defined as the number of gram equivalents of the solute dissolved per litre (or cubic decimeter) of the solution. It is denoted by the symbol 'N' and its unit is equivalent per litre (eq/L).
@$\begin{align*}\text{Normality (N)} = \frac{\text{Number of gram equivalents of solute}}{\text{Volume of the solution in litres}}\end{align*}@$
The number of gram equivalents of a solute is calculated by dividing the mass of the solute by its equivalent mass:
@$\begin{align*}\text{Number of gram equivalents of solute} = \frac{\text{Mass of solute (in grams)}}{\text{Equivalent mass of solute (in g/eq)}}\end{align*}@$
Combining these, if 'w' grams of a solute with an equivalent mass 'Eq. mass' are dissolved in 'V' mL of solution, the normality can be calculated as:
@$\begin{align*}\text{N} = \frac{w}{\text{Eq. mass}} \times \frac{1000}{V}\end{align*}@$
Equivalent Mass of a Substance
The equivalent mass of a substance depends on the chemical reaction it undergoes. Some common definitions are:
Element: The mass of the element that combines with or displaces 1.008 parts by mass of hydrogen, 8 parts by mass of oxygen, or 35.5 parts by mass of chlorine. For metals and other elements, the equivalent mass can be calculated using the following formula:
@$\begin{align*}\text{Eq. mass of an element} = \frac{\text{Atomic mass of the element}}{\text{Valency of the element}}\end{align*}@$
Acid: The molar mass of the acid divided by its basicity (the number of displaceable H+ ions per molecule) is the equivalent mass of an acid.
@$\begin{align*}\text{Eq. mass of an acid} = \frac{\text{Molar mass of the acid}}{\text{Basicity of the acid}}\end{align*}@$
For example, for HCl, basicity = 1; for H2SO4, basicity = 2; for H3PO4, basicity = 3.
Base: The molar mass of the base divided by its acidity (the number of displaceable OH- ions per molecule) is the equivalent mass of a base.
@$\begin{align*}\text{Eq. mass of a base} = \frac{\text{Molar mass of the base}}{\text{Acidity of the base}}\end{align*}@$
For example, for NaOH, acidity = 1; for Ca(OH)2, acidity = 2; for Al(OH)3, acidity = 3.
Salt: The molar mass of the salt divided by the total positive valency of the metal atoms is the equivalent mass of a salt.
@$\begin{align*}\text{Eq. mass of a salt} = \frac{\text{Molar mass of the salt}}{\text{Total positive valency of the metal atoms}}\end{align*}@$
For example, in Al2(SO4)3, the total positive valency of aluminum is 2 x (+3) = +6.
Ion: The formula mass of the ion divided by the magnitude of the charge on the ion is the equivalent mass of an ion.
@$\begin{align*}\text{Eq. mass of an ion} = \frac{\text{Formula mass of the ion}}{\text{Charge on the ion}}\end{align*}@$
For example, for SO42-, the equivalent mass is its formula mass divided by 2.
Oxidizing/Reducing Agent: The molar mass or atomic mass divided by the number of electrons lost or gained by one molecule or atom of the substance in a redox reaction is the equivalent mass of the substance.
Relationship between Normality and Molarity
The normality of a solution is related to its molarity by the following equation:
@$\begin{align*}\text{Normality} = \text{Molarity} \times \frac{\text{Molar mass}}{\text{Equivalent mass}}\end{align*}@$
For an acid:
@$\begin{align*}\frac{\text{Molar mass}}{\text{Equivalent mass}} = \text{Basicity}\end{align*}@$
@$\begin{align*}\text{Normality of an acid} = \text{Molarity} \times \text{Basicity}\end{align*}@$
For a base:
@$\begin{align*}\frac{\text{Molar mass}}{\text{Equivalent mass}} = \text{Acidity}\end{align*}@$
@$\begin{align*} \text{Normality of a base} = \text{Molarity} \times \text{Acidity}\end{align*}@$
For example, a 0.5 M solution of H2SO4 will have a normality of 0.5 M x 2 = 1 N.
Common Fractions of Molarity and Normality
- 1 M (or 1 N) = Molar (or Normal)
- M/2 (or N/2) = Semimolar (or Seminormal)
- M/10 (or N/10) = Decimolar (or Decinormal)
- M/100 (or N/100) = Centimolar (or Centinormal)
- M/1000 (or N/1000) = Millimolar (or Millinormal)
Examples of Normality
Example 1:
Calculate the normality of a solution containing 0.49 grams of sulfuric acid (H2SO4) dissolved in 250 cm3 of solution.
The equivalent mass of sulfuric acid is its molar mass (98 g/mol) divided by its basicity (2, since it has two replaceable H+ ions), which is 98/2 = 49 g/eq.
@$\begin{align*}\text{Eq. mass of} \ \ \text{H}_2\text{S}\text{O}_4 = \frac{98}{2} = 49 \ \ \text{g/eq}\end{align*}@$
@$\begin{align*}\text{N} = \frac{0.49 \text{ g}}{49 \text{ g/eq}} \times \frac{1000 \, mL/L}{250 \text{ mL}} = 0.01 \text{ eq} \times 4 \text{ L}^{-1} = 0.04 \text{ eq L}^{-1} = 0.04 \text{ N}\end{align*}@$
Example 2:
Calculate the normality of a solution prepared by dissolving 8.0 grams of sodium hydroxide (NaOH) in enough water to make a final volume of 500 ml. (Molar mass of NaOH = 40 g/mol, Acidity of NaOH = 1)
To calculate the normality, find the number of gram equivalents of the solute (NaOH) and divide it by the volume of the solution in litres.
Step 1: Calculate the equivalent mass of NaOH
The equivalent mass of a base is given by:
@$\begin{align*}\text{Equivalent mass of base} = \frac{\text{Molar mass of base}}{\text{Acidity of base}}\end{align*}@$
For NaOH, the molar mass is 40 g/mol and the acidity is 1 (because it has one replaceable hydroxide ion).
@$\begin{align*}\text{Equivalent mass of NaOH} = \frac{40 \text{ g/mol}}{1} = 40 \text{ g/eq}\end{align*}@$
Step 2: Calculate the number of gram equivalents of NaOH.
The number of gram equivalents is calculated by dividing the mass of the solute by its equivalent mass:
@$\begin{align*}\text{Number of gram equivalents} = \frac{\text{Mass of solute}}{\text{Equivalent mass of solute}}\end{align*}@$
@$\begin{align*}\text{Number of gram equivalents of NaOH} = \frac{8.0 \text{ g}}{40 \text{ g/eq}} = 0.2 \text{ eq}\end{align*}@$
Step 3: Convert the volume of the solution to liters.
The final volume of the solution is given as 500 ml. To convert this to litres, we divide by 1000:
@$\begin{align*}\text{Volume of solution in liters} = \frac{\text{Volume in ml}}{1000}\end{align*}@$
@$\begin{align*}\text{Volume of solution in litres} = \frac{500 \text{ ml}}{1000 \text{ ml/L}} = 0.5 \text{ L}\end{align*}@$
Step 4: Calculate the normality of the solution.
Normality (N) is defined as the number of gram equivalents of solute per liter of solution:
@$\begin{align*}\text{Normality (N)} = \frac{\text{Number of gram equivalents of solute}}{\text{Volume of the solution in litres}}\end{align*}@$
@$\begin{align*}\text{Normality (N)} = \frac{0.2 \text{ eq}}{0.5 \text{ L}} = 0.4 \text{ eq/L}\end{align*}@$
Therefore, the normality of the sodium hydroxide solution is0.4 N.
Molality (m)
Molality is defined as the number of moles of the solute dissolved in 1000 grams (or 1 kg) of the solvent. It is denoted by the symbol 'm' and its unit is mol/kg.
@$\begin{align*}\text{Molality (m)} = \frac{\text{Number of moles of the solute}}{\text{Mass of the solvent in kilograms}}\end{align*}@$
If 'a' grams of the solute (with molar mass 'Molar mass') are dissolved in 'b' grams of the solvent, then the molality is:
@$\begin{align*}m = \frac{a}{\text{Molar mass}} \times \frac{1000}{b}\end{align*}@$
Examples of Molality
Example 1:
Calculate the molality of a solution containing 1.325 grams of anhydrous sodium carbonate (Na2CO3, molar mass = 106 g/mol) dissolved in 250 grams of water.
@$\begin{align*}\text{Molality (m)} = \frac{\text{Number of moles of the solute}}{\text{Mass of the solvent in kilograms}}\end{align*}@$
@$\begin{align*}m = \frac{1.325 \text{ g}}{106 \text{ g/mol}} \times \frac{1000 \, g/kg}{250 \text{ g}} = 0.0125 \text{ mol} \times 4 \text{ kg}^{-1} = 0.05 \text{ mol kg}^{-1} = 0.05 \text{ m}\end{align*}@$
Mole Fraction (x)
The mole fraction of a component in a solution is the ratio of the number of moles of that component to the total number of moles of all the components in the solution. For a solution containing a solute (component 2) dissolved in a solvent (component 1), the mole fractions are:
@$\begin{align*}\text{Mole fraction of solvent } (x_1) = \frac{n_1}{n_1 + n_2}\end{align*}@$
@$\begin{align*}\text{Mole fraction of solute } (x_2) = \frac{n_2}{n_1 + n_2}\end{align*}@$
where n1 is the number of moles of the solvent and n2 is the number of moles of the solute.
The sum of the mole fractions of all the components in a solution is always equal to unity. Mole fraction is a dimensionless quantity.
For example, if a solution contains 4 moles of ethanol and 6 moles of water, then:
@$\begin{align*}\text{Mole fraction of ethanol } (x_{\text{ethanol}}) = \frac{4}{4 + 6} = \frac{4}{10} = 0.4\end{align*}@$
@$\begin{align*}\text{Mole fraction of water } (x_{\text{water}}) = \frac{6}{4 + 6} = \frac{6}{10} = 0.6\end{align*}@$
@$\begin{align*}x_{\text{ethanol}} + x_{\text{water}} = 0.4 + 0.6 = 1\end{align*}@$
Examples of Mole Fraction
Example 1:
Calculate the mole fraction of urea (NH₂CONH₂) in a solution prepared by dissolving 6 g of urea (molar mass = 60 g/mol) in 94 g of water (molar mass = 18 g/mol).
@$\begin{align*}\text{Moles of urea} = \frac{6}{60} = 0.1\ \text{mol}\end{align*}@$
@$\begin{align*}\text{Moles of water} = \frac{94}{18} \approx 5.22\ \text{mol}\end{align*}@$
@$\begin{align*}\text{Total moles} = 0.1 + 5.22 = 5.32\end{align*}@$
@$\begin{align*}\text{Mole fraction of urea} = \chi_{\text{urea}} = \frac{0.1}{5.32} \approx 0.0188\end{align*}@$
@$\begin{align*}\text{Mole fraction of water} = \chi_{\text{water}} = \frac{5.22}{5.32} \approx 0.9812\end{align*}@$
Example 2:
A solution is made by mixing 78 g of benzene (C₆H₆) with 92 g of toluene (C₇H₈). Calculate the mole fractions of benzene and toluene in the mixture.
(Molar masses: benzene = 78 g/mol, toluene = 92 g/mol)
@$\begin{align*}\text{Moles of benzene} = \frac{78}{78} = 1\ \text{mol}\end{align*}@$
@$\begin{align*}\text{Moles of toluene} = \frac{92}{92} = 1\ \text{mol}\end{align*}@$
@$\begin{align*}\text{Total moles} = 1 + 1 = 2\end{align*}@$
@$\begin{align*}\text{Mole fraction of benzene} = \chi_{\text{benzene}} = \frac{1}{2} = 0.5\end{align*}@$
@$\begin{align*}\text{Mole fraction of toluene} = \chi_{\text{toluene}} = \frac{1}{2} = 0.5\end{align*}@$
Mass Fraction
The mass fraction of a component is defined as the mass of that component divided by the total mass of the solution. For a binary solution containing components A and B with masses wA and wB respectively, the mass fractions are:
@$\begin{align*}\text{Mass fraction of A } (X_A) = \frac{w_A}{w_A + w_B}\end{align*}@$
@$\begin{align*}\text{Mass fraction of B } (X_B) = \frac{w_B}{w_A + w_B}\end{align*}@$
The sum of the mass fractions of all components in a solution is always equal to unity. Mass fraction is also a dimensionless quantity. Multiplying the mass fraction by 100 gives the mass percentage.
For example, if 20 g of substance A is mixed with 80 g of substance B, then:
@$\begin{align*}\text{Mass fraction of A } (X_A) = \frac{20}{20 + 80} = \frac{20}{100} = 0.2\end{align*}@$
@$\begin{align*}\text{Mass fraction of B } (X_B) = \frac{80}{20 + 80} = \frac{80}{100} = 0.8\end{align*}@$
@$\begin{align*}X_A + X_B = 0.2 + 0.8 = 1\end{align*}@$
@$\begin{align*}\text{The mass percentage of A} = 0.2 \times 100 = 20 \%\end{align*}@$
@$\begin{align*}\text{The mass percentage of B} = 0.8 \times 100 = 80\%\end{align*}@$
Examples of Mass Fraction
Example 1:
A solution is prepared by dissolving 10 g of sodium chloride (NaCl) in 90 g of water. Calculate the mass fraction of NaCl and water in the solution.
@$\begin{align*}\text{Total mass of the solution} = 10 \text{g} \ \ \text{(NaCl)} + 90 \text{g} \ \ \text{(water)} = 100 g\end{align*}@$
@$\begin{align*}\text{Mass fraction of NaCl} = \frac{10}{100} = 0.10\end{align*}@$
@$\begin{align*}\text{Mass fraction of water} = \frac{90}{100} = 0.90\end{align*}@$
Example 2:
A solution contains 30 g of ethanol (C₂H₅OH) mixed with 120 g of water. Calculate the mass fraction of ethanol and water in the solution.
@$\begin{align*}\text{Total mass of the solution} = 30 \text{g (ethanol)} + 120 \text{g (water)} = 150 g\end{align*}@$
@$\begin{align*}\text{Mass fraction of ethanol} = \frac{30}{150} = 0.20\end{align*}@$
@$\begin{align*}\text{Mass fraction of water} = \frac{120}{150} = 0.80\end{align*}@$
Parts per Million (ppm)
For very dilute solutions, where the amount of solute is very small compared to the amount of solution, the concentration is often expressed in parts per million (ppm). It is defined as the mass of the solute present in one million (106) parts by mass of the solution.
@$\begin{align*}\text{ppm of solute A} = \frac{\text{Mass of solute A}}{\text{Mass of solution}} \times 10^6\end{align*}@$
In the case of gaseous pollutants in the atmosphere, ppm is often expressed in terms of volume:
@$\begin{align*}\text{ppm by volume of a gas} = \frac{\text{Volume of the gas}}{\text{Total volume of air}} \times 10^6\end{align*}@$
For example, if 10 mg of a salt is dissolved in 1 kg of water, the concentration of the salt is:
@$\begin{align*}\text{ppm} = \frac{10 \times 10^{-3} \text{ g}}{1000 \text{ g}} \times 10^6 = 10 \text{ ppm}\end{align*}@$
Examples of Parts per Million
Example 1:
A sample of drinking water contains 0.005 g of fluoride ions in 1 litre of water. Calculate the concentration of fluoride in ppm. (Note: 1 litre of water ≈ 1000 g)
@$\begin{align*}\text{Mass of solute (fluoride)} = 0.005 \text{g}\end{align*}@$
@$\begin{align*}\text{Mass of solution (water)} = 1000 \text{g}\end{align*}@$
@$\begin{align*}\text{ppm} = \frac{\text{Mass of solute}}{\text{Mass of solution}} \times 10^6 = \frac{0.005}{1000} \times 10^6 = 5\ \text{ppm}\end{align*}@$
Example 2:
A 2.5 kg soil sample is found to contain 0.002 g of lead (Pb). Calculate the concentration of lead in ppm.
@$\begin{align*}\text{Mass of solute (lead)} = 0.002 \text{g}\end{align*}@$
@$\begin{align*}\text{Mass of solution (soil)} = 2.5 \text{kg} = 2500 \text{g}\end{align*}@$
@$\begin{align*}\text{ppm} = \frac{0.002}{2500} \times 10^6 = 0.8\ \text{ppm}\end{align*}@$
Temperature Dependence of Concentration Units
It is important to note that some concentration units are temperature-dependent, while others are not. Concentration units involving volume (like molarity, normality, percentage by volume, and mass/volume percentage) change with temperature because the volume of a solution changes with temperature due to the expansion or contraction of liquids.
On the other hand, concentration units based on mass (like percentage by mass, molality, mole fraction, and mass fraction) do not change with temperature because mass remains constant regardless of temperature. For this reason, molality, mole fraction, and mass fraction are often preferred over molarity and normality when studying phenomena that occur over a range of temperatures.
Relationships Between Volume and Normality or Molarity
When a solution is diluted by adding more solvent, the amount of solute remains the same, but the volume of the solution increases, leading to a decrease in concentration. We can use specific equations to calculate the new normality or molarity after dilution.
Normality Equation
If a solution with an initial volume V1 and normality N1 is diluted to a final volume V2 and the new normality is N2, the following relationship holds:
@$\begin{align*}N_1 \times V_1 = N_2 \times V_2\end{align*}@$
This equation is based on the principle that the number of gram equivalents of the solute remains constant during dilution.
Similarly, if a volume V1 of a solution with normality N1 reacts exactly with a volume V2 of another solution with normality N2, the same equation applies, as the number of gram equivalents of the reacting species must be equal at the equivalence point.
Molarity Equation
Analogously, for molarity, if a solution with an initial volume V1 and molarity M1 is diluted to a final volume V2 with a new molarity M2, the relationship is:
@$\begin{align*}M_1 \times V_1 = M_2 \times V_2\end{align*}@$
This equation is based on the conservation of the number of moles of the solute during dilution.
However, when studying the reaction between two solutions in terms of their molarities, the stoichiometry of the balanced chemical equation must be considered. The molarity equation for the reaction between two reactants is:
@$\begin{align*}\frac{M_1 \times V_1}{n_1} = \frac{M_2 \times V_2}{n_2}\end{align*}@$
@$\begin{align*}n_2 \times M_1 \times V_1 = n_1 \times M_2 \times V_2\end{align*}@$
@$\begin{align*}N_1 \times V_1 = N_2 \times V_2\end{align*}@$
where n1 is the number of moles of reactant 1 and n2 is the number of moles of reactant 2 as given by the balanced chemical equation.
Mixing Solutions of the Same Solute and Same Solvent
Mixing Solutions with the Same Normality or Molarity: If a volume V1 of a solution with normality N1 is mixed with a volume V2 of another solution containing same solute and solvent with normality N2, the normality N3 of the final solution can be calculated as:
@$\begin{align*}N_3 = \frac{N_1 V_1 + N_2 V_2}{V_1 + V_2}\end{align*}@$
In case of further dilution of solution by water, normality N3:
@$\begin{align*}N_3 = \frac{N_1 V_1 + N_2 V_2}{V_1 + V_2 + V_\text{water}}\end{align*}@$
A similar equation applies for molarity:
@$\begin{align*}M_3 = \frac{M_1 V_1 + M_2 V_2}{V_1 + V_2}\end{align*}@$
Mixing Acidic and Basic Solutions: When a solution of an acid is mixed with a solution of a base, neutralization occurs. To find the normality of the resulting solution:
1. Calculate the number of gram equivalents of the acid and the base.
2. The smaller number of gram equivalents will neutralize an equal number of gram equivalents of the other.
3. The number of gram equivalents of the excess reactant left is the difference between the initial gram equivalents of the acid and the base.
4. The normality of the final solution is the number of gram equivalents of the excess reactant divided by the total volume of the solution in litres.
Remember the relationships between normality and molarity for acids and bases:
@$\begin{align*}\text{Normality of acid} = \text{Basicity} \times \text{Molarity}\end{align*}@$
@$\begin{align*}\text{Normality of base} = \text{Acidity} \times \text{Molarity}\end{align*}@$
Examples of Relationships Between Volume and Normality or Molarity
Example 1:
50 ml of a hydrochloric acid (HCl) solution of normality 4 N is diluted to 250 ml. Calculate the new normality of the diluted solution.
@$\begin{align*}N_1 \times V_1 = N_2 \times V_2\end{align*}@$
Substitute values:
@$\begin{align*}4 \times 50 = N_2 \times 250\end{align*}@$
@$\begin{align*}N_2 = \frac{4 \times 50}{250} = \frac{200}{250} = 0.8\ N\end{align*}@$
Example 2:
25 mL of 1.0 M H₂SO₄ is required to completely neutralize 50 ml of NaOH solution. Calculate the molarity of NaOH, given that the basicity of H₂SO₄ is 2.
For neutralisation reaction, number of equivalents of H2SO4 = number of equivalents of base
@$\begin{align*}N_1V_1 = N_2V_2\end{align*}@$
@$\begin{align*}n_1 \times M_1 \times V_1 = n_2 \times M_2 \times V_2\end{align*}@$
@$\begin{align*}M_1 = 1.0\ M,\ V_1 = 25\ ml,\ n_1 = 2 \ \ \text{(basicity of H₂SO₄)}\end{align*}@$
@$\begin{align*}V_2 = 50\ ml,\ n_2 = 1 \text{(acidity of NaOH)}\end{align*}@$
Let M2 be the molarity of NaOH.
@$\begin{align*}2 \times 1.0 \times 25 = 1 \times M_2 \times 50\end{align*}@$
@$\begin{align*}M_2 = \frac{50}{50} = 1.0\ M\end{align*}@$
The molarity of the NaOH solution is 1.0 M.
Example 3:
100 ml of HCl solution of normality 2 N is mixed with 300 ml of another HCl solution of normality 0.5 N. Calculate the normality of the resulting solution.
Use the mixing formula:
@$\begin{align*}N_3 = \frac{N_1 V_1 + N_2 V_2}{V_1 + V_2}\end{align*}@$
@$\begin{align*}N_3 = \frac{(2 \times 100) + (0.5 \times 300)}{100 + 300} = \frac{200 + 150}{400} = \frac{350}{400} = 0.875\ N\end{align*}@$
The normality of the final solution is 0.875 N.
Example 4:
100 ml of 1.0 N HCl is mixed with 60 ml of 2.0 N NaOH. Calculate the normality of the resulting solution after neutralization.
Calculate gram equivalents:
@$\begin{align*}\text{Gram equivalents of HCl} = N \times V = 1.0 \times \frac{100}{1000} = 0.1\end{align*}@$
@$\begin{align*}\text{Gram equivalents of NaOH} = 2.0 \times \frac{60}{1000} = 0.12 \ \ \text{eq}\end{align*}@$
@$\begin{align*}\text{Excess gram equivalents of NaOH} = \text{0.12 eq − 0.1 eq} = \text{0.02 eq}\end{align*}@$
@$\begin{align*}\text{The total volume of resulting solution} = 100 + 60 = \text{160 ml}\end{align*}@$
@$\begin{align*}\text{Normality of resulting solution} = \frac{0.02}{0.160} = \text{0.125 N}\end{align*}@$
The resulting solution is basic due to excess of NaOH and its normality is 0.125 N.
Case Study: Quantitative Preparation and Analysis of Pharmaceutical Solutions Using Concentration Units
In a pharmaceutical laboratory, a team is working on preparing intravenous (IV) glucose solutions for diabetic patients. They prepare a 5% (w/v) glucose solution by dissolving 5 grams of glucose (C6H12O6) in enough water to make the final volume 100 ml. For more concentrated dosages, they also prepare a 10% (w/v) solution. Meanwhile, in the biochemistry section, a saline solution of 0.9% (w/v) NaCl is prepared for injections. A chemist is tasked with preparing 0.5 M NaCl by dissolving the appropriate amount of salt (molar mass = 58.5 g/mol) in water to make 1 L of solution. Another analyst calculates the mole fraction of ethanol in a solution made by mixing 46 g ethanol (molar mass = 46 g/mol) with 180 g water (molar mass = 18 g/mol). They also compare molarity and molality to determine which is affected by temperature and which is independent. In the quality check section, they use ppm concentration to detect impurities present in microgram levels per liter of sample.
Video: Concentration of Solution Formulas
| Summary of Expressing Concentration of Solutions |
|
Review Questions on Expressing Concentration of Solutions
- A solution is prepared by dissolving 15 grams of glucose (C6H12O6) in 150 grams of water. Calculate the percentage by mass of glucose in the solution.
- What is the molarity of a solution containing 20 grams of sodium hydroxide (NaOH) dissolved in 2.0 liters of solution? (Molar mass of NaOH = 40 g/mol)
- Calculate the molality of a solution prepared by dissolving 0.5 moles of urea (NH2CONH2) in 250 grams of water.
- If 250 mL of a 0.2 M hydrochloric acid (HCl) solution is diluted with water to a final volume of 500 mL, what is the molarity of the resulting solution?
- A solution contains 2 moles of ethanol and 8 moles of water. What is the mole fraction of ethanol in the solution?
- What volume of a 0.8 M sodium hydroxide (NaOH) solution is required to completely neutralize 40 ml of a 0.2 M sulfuric acid (H2SO4) solution?
- 200 mL of a 0.5 N hydrochloric acid (HCl) solution is mixed with 300 mL of a 0.1 N nitric acid (HNO3) solution. Assuming the volumes are additive and the acids do not react with each other, what is the normality of the resulting acidic solution?
- You have 100 mL of a 1.5 M solution of glucose. You need to prepare 500 mL of a 0.3 M glucose solution for an experiment. What volume of the 1.5 M stock solution should you take and how much water should you add to achieve the desired concentration?
| Image | Reference | Attributions |
|---|