Acid Rain
Lesson

Why is this gargoyle worn out?
The construction of Notre Dame Cathedral in Paris, France took nearly 200 years, beginning in 1163 and completing in 1345. Much of the original masonry and decorations, like this gargoyle, were carved from limestone. However, in the 18th and 19th century, increased air pollution in Paris led to acid rain, which caused the stone to weather much more quickly. Limestone structures around the world are vulnerable to acid rain, which has become more common due to pollutants from fossil fuel burning.
Acid Rain
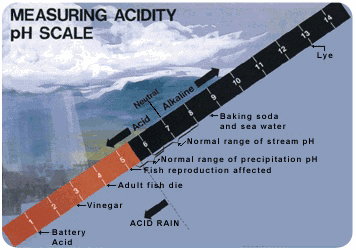
Acid rain is rain that has a pH less than 5 (Figure below). Acidity is measured on the pH scale. Lower numbers are more acidic, and higher numbers are less acidic (also called more alkaline). An acid has a pH of less than 7. The pH of normal rain is 5.6. It’s slightly acidic because carbon dioxide in the air dissolves in rain. This forms carbonic acid, a weak acid.
How Acid Rain Forms
Nitrogen and sulfur oxides are released when fossil fuels are burned. They gases can move downwind and don’t come out of the atmosphere until they dissolve in rainwater (Figure below). This forms nitric and sulfuric acids. Both are strong acids. Acid rain with a pH as low as 4.0 is now common in many areas. Acid fog may be even more acidic than acid rain. Fog with a pH as low as 1.7 has been recorded. The acids in precipitation are diluted by water, so the damage isn’t immediately noticeable.

Effects of Acid Rain
The image below shows some of the damage done by acid rain (Figure below). Acid rain ends up in soil and bodies of water. This can make them very acidic. The acid strips soil of its nutrients. These changes can kill trees, fish, and other living things. Acid rain also dissolves limestone and marble. This can damage buildings, monuments, and statues.

Summary
- Nitrogen and sulfur compounds emitted high into the atmosphere create acids. These acids may fall as acid rain.
- Acidity is measured on a pH scale. Rain that is 5.0 or less on that scale is considered acid rain.
- Acid rain weakens plants and animals and damages cultural treasures.
Review
- Why does acid rain do a lot of damage far from where the smoke was released?
- What damage does acid rain do to organisms? What damage does it do to cultural structures?
- Describe the pH scale. What can you say about a liquid with a pH of 2.5? What about a liquid with a pH of 10.5?
Explore More
Use the resource below to answer the questions that follow.
- What causes acid rain?
- What two gases react with water to make acid rain?
- Where relative to where the gases are emitted does acid rain strike?
- What damage can acid rain cause?
- How can we reduce acid rain?
- What are scrubbers?
- What is slurry?
- How effective are scrubbers?
Asked by Students
Overview
- Nitrogen and sulfur compounds emitted high into the atmosphere create acids. These acids may fall as acid rain.
- Acidity is measured on a pH scale. Rain that is 5.0 or less on that scale is considered acid rain.
- Acid rain weakens plants and animals and damages cultural treasures.
Notable Images
Vocabulary
Test Your Knowledge
Natural rain has a pH of _________________.
8.6
5.6
7.0
4.0
Acid rain is rain that has a pH less than 5 (Figure here). Acidity is measured on the pH scale. Lower numbers are more acidic, and higher numbers are less acidic (also called more alkaline). An acid has a pH of less than 7. The pH of normal rain is 5.6. It’s slightly acidic because carbon dioxide in the air dissolves in rain. This forms carbonic acid, a weak acid.

What is another form of acidic precipitation that can have an even lower pH than acid rain?
acid fog
acid hail
acid sleet
acid snow
Acid rain forms when nitrogen and sulfur oxides in air dissolve in rain (Figure here). This forms nitric and sulfuric acids. Both are strong acids. Acid rain with a pH as low as 4.0 is now common in many areas. Acid fog may be even more acidic than acid rain. Fog with a pH as low as 1.7 has been recorded. That’s the same pH as toilet bowl cleaner!

Study Guide
Asked by Students
Related Content
| Image | Reference | Attributions |
|---|---|---|
 | Credit: Flickr:Chadica Source: http://www.flickr.com/photos/chadica/3502986841/ License: CC BY 2.0 | |
 | Credit: Courtesy of the US Geological Survey Source: http://pubs.usgs.gov/gip/acidrain/1.html License: Public Domain | |
 | Credit: Courtesy of the U.S. EPA Source: http://www.epa.gov/bioiweb1/aquatic/airpoll.html License: Public Domain | |
 | Credit: User:Npik/Wikimedia Commons Source: http://commons.wikimedia.org/wiki/File:Acid_rain_woods1.JPG License: Public Domain |